|
For a first-order reaction, the half-life is independent of concentration and constant over time. The half-life of a reaction is the time required for a reactant to reach one-half its initial concentration or pressure. 5,730 years The graph shows that 50 percent of the carbon-14 atoms have decayed after 5,730 years, so 5,730 is the half-life of carbon-14. What is the half-life of carbon-14 when 50% remains how many years have passed?ġ. The half-life of potassium-40 that decays through beta emission is 1.28 × 109 years, however the half-life of potassium-40 that decays through positron emission is 1.19 × 1010 years. The beta decay/electron capture process occurs about 10 times faster than the positron decay process. For example, the half-life of carbon-14 is 5,730 years. 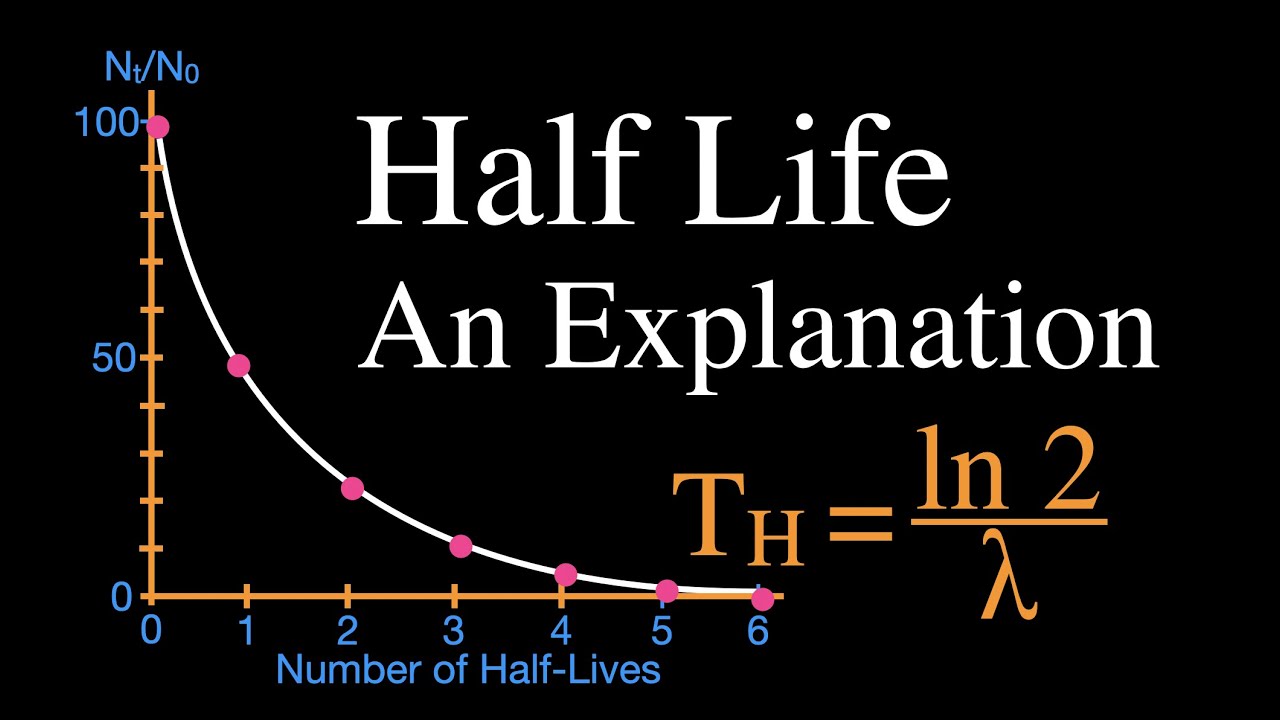
The half-life is the time it takes for half of a given amount of an isotope to decay. 
What is the half-life of the 10 atoms of carbon-14?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
